(VSM) Redefining Value Stream Mapping for the Regulated MedTech Environment
Value Stream Mapping has long been a cornerstone of Lean Manufacturing. It visualizes material and information flow from raw material to finished product. But in MedTech, traditional Value Stream Mapping tells only part of the story.
THE LEARNING LOOPINTELLIGENT MANUFACTURING TRANSFORMATION
Manfred Maiers
3/5/20265 min read


Redefining Value Stream Mapping for the Regulated MedTech Environment
Operational Excellence in medical device manufacturing has always required more than speed and cost control. In regulated industries, flow efficiency is only one dimension of performance. The true measure of excellence is the ability to deliver safe, compliant, high-quality products consistently, under increasing regulatory scrutiny and rising product complexity.
Value Stream Mapping (VSM) has long been a cornerstone of Lean Manufacturing. It visualizes material and information flow from raw material to finished product. It highlights cycle time, takt time, lead time, changeover, and bottlenecks. It exposes waiting, overproduction, and excess inventory. It is powerful, intuitive, and operationally effective.
But in MedTech, traditional Value Stream Mapping tells only part of the story.
The Foundations of Value Stream Mapping
Value Stream Mapping originated within Lean production systems as a structured way to visualize how value flows through a process. A typical VSM captures:
Process steps and flow.
Cycle time and takt alignment.
Changeover time.
Work in process inventory.
Lead time across the stream.
Information flows between planning and production.
When executed properly, VSM creates clarity. It reveals non-value-added activities and enables targeted improvement events. Leadership can quickly identify bottlenecks and systemic inefficiencies.
However, traditional VSM is designed for throughput and waste elimination. It was not originally designed for industries where regulatory compliance and risk management are foundational constraints.
In MedTech manufacturing, this omission becomes significant.
The Structural Blind Spot: Quality and Regulatory Intelligence
A standard VSM process box is rich with efficiency metrics but typically silent on quality performance. It may display cycle time and uptime, yet it does not show:
Defect rates.
Scrap percentages.
Rework loops.
Inspection escape rates.
Nonconformance trends.
CAPA signals.
Audit observations linked to specific steps.
The result is a structural blind spot.
A process step may appear lean and efficient on paper while quietly generating rework or feeding downstream inspection failures. Leadership sees flow but not risk concentration. Operations sees throughput but not regulatory exposure.
In a regulated environment governed by ISO 13485, FDA QMSR expectations, and EU MDR requirements, that separation is not sustainable. Efficiency without quality visibility is incomplete. And compliance without operational context is reactive.
Operational Excellence in MedTech requires integration.
From VSM to Enhanced Value Stream Mapping
The next evolution is the Enhanced Value Stream Map, or EVSM.
At its core, EVSM retains the clarity of traditional flow visualization. But it embeds quality intelligence directly inside each process box.
Instead of only displaying cycle time, a process step now includes:
First Pass Yield.
First Time Yield.
Scrap rate.
Rework rate.
Process capability indicators.
Inspection effectiveness metrics.
Trend indicators for nonconformance.
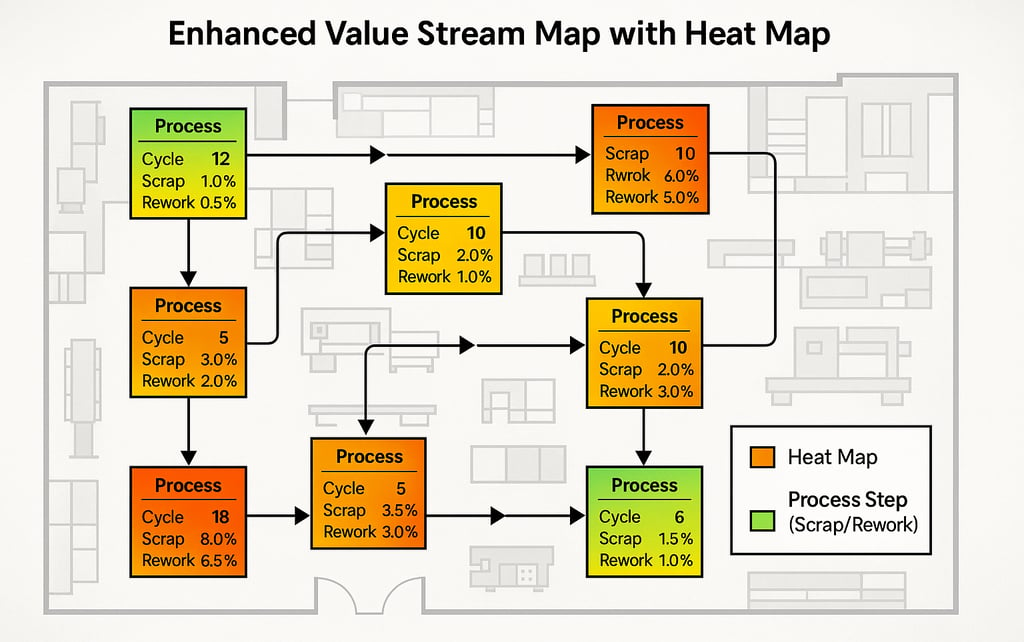
Immediately, the map transforms from a flow diagram into a performance intelligence system.
Patterns that were previously hidden become visible. A process that runs fast but generates downstream inspection failures becomes a risk hotspot. A shared machine across value streams reveals systemic quality variation. A step with marginal yield exposes a leverage point for both cost and compliance improvement.
When overlaid on a plant layout, EVSM can be visualized as a heat map of operational risk and quality loss. Areas with elevated scrap or rework concentrations are no longer anecdotal. They are structurally mapped.
The conversation shifts from isolated firefighting to systemic optimization.
Integrating Regulatory Goals and Risk-Based KPIs
In MedTech, Operational Excellence must align with regulatory goals. That alignment requires translating regulatory expectations into measurable, stream-level KPIs.
Consider what regulators expect:
Demonstrated process control.
Objective evidence of risk mitigation.
Traceability of nonconformance to root cause.
Continuous improvement based on data.
Risk-based decision making.
These are not abstract requirements. They can be mapped.
An EVSM allows each process step to link to:
Associated PFMEA failure modes.
Occurrence data derived from real scrap and defect rates.
Detection effectiveness based on inspection outcomes.
Control plan elements tied to risk mitigation.
Historical CAPA triggers.
When quality metrics feed directly into PFMEA Occurrence and Detection ratings, the value stream becomes part of a closed-loop risk management architecture.
Risk is no longer static, defined only during product development. It becomes dynamic, informed by production reality.
This integration creates a structural bridge between Lean tools and regulatory risk frameworks. Historically, these domains have operated in silos. EVSM dissolves that boundary.
A Conceptual Model: Flow + Risk + Compliance
Traditional Lean thinking focuses on flow.
Regulatory systems focus on compliance.
Risk management frameworks focus on hazard mitigation.
Intelligent Operational Excellence needs the convergence of all three.
Flow + Risk + Compliance = Intelligent Operational Excellence
Flow ensures efficiency and responsiveness.
Risk visibility ensures patient safety and proactive mitigation.
Compliance ensures sustainability under regulatory oversight.
When quality KPIs, regulatory objectives, and risk metrics are embedded directly into the value stream visualization, operational decisions become multidimensional.
A throughput improvement initiative is evaluated not only for cycle time reduction, but also for its effect on risk profile and detection robustness. A process automation investment is justified not only by labor savings, but by improved yield stability and reduced regulatory exposure.
This integrated perspective elevates Operational Excellence from tactical improvement to strategic governance.
Beyond Reactive CAPA Management
Many organizations operate reactively. A nonconformance spike triggers investigation. A complaint trend initiates a CAPA. An audit observation results in corrective action.
While necessary, this reactive model is structurally limited.
EVSM enables proactive monitoring.
If a specific process step shows declining First Pass Yield, that signal feeds into risk review before customer impact occurs. If inspection failures cluster around a shared workstation, leadership can assess detection strategy and training adequacy before systemic issues escalate.
The map becomes an early warning system.
Instead of asking, “Why did this happen?” organizations begin asking, “Where is risk accumulating?”
That shift represents maturity.
Strategic Implications for MedTech Leaders
Embedding regulatory and quality intelligence into the value stream has several strategic consequences.
First, it strengthens audit readiness. When an auditor asks how process performance links to risk controls, the answer is not buried in separate documents. The relationship is visible in the operational model.
Second, it improves decision making. Leadership discussions move beyond isolated KPIs. Tradeoffs between speed, cost, and risk become transparent.
Third, it sharpens continuous improvement. Kaizen events can target areas with the highest combined impact on yield, compliance, and throughput. Resources are allocated based on systemic data rather than anecdotal urgency.
Fourth, it improves patient safety outcomes. Process risks are identified earlier. Control weaknesses are detected faster. The connection between manufacturing variation and clinical consequence becomes clearer.
In a world of increasing product complexity, tighter global regulations, and cost pressure, this integrated approach is not optional. It is structural.
The Future of Value Stream Mapping in Regulated Manufacturing
Value Stream Mapping was never meant to be static. Like all Lean tools, it evolves with context.
In MedTech, that evolution must reflect regulatory reality. Efficiency alone does not define excellence. Compliance alone does not ensure competitiveness. Risk management alone does not guarantee flow stability.
The next generation of Operational Excellence requires structured visibility across all dimensions of performance.
Enhanced Value Stream Mapping is not a cosmetic upgrade. It is a transformation of how organizations see their operations. It makes quality measurable within flow. It makes regulatory objectives operational. It makes risk dynamic rather than theoretical.
Intelligent Operational Excellence begins when quality and compliance are no longer external overlays, but embedded properties of the value stream itself.
When quality becomes visible at the same level as cycle time, Operational Excellence is no longer just Lean.


It becomes intelligent.
