Risk Management at the Edge of Uncertainty
Safe Launch for MedTech Why Product Launch and Product Change Demand a Different Playbook
THE LEARNING LOOPRISK MANAGEMENT
Manfred Maiers
2/3/20265 min read


Risk Management at the Edge of Uncertainty: Safe Launch for MedTech
Why Product Launch and Product Change Demand a Different Playbook
In the earlier articles of this series, we showed the Risk Management Plan as the recipe and the Risk Management File as the living record that proves risk has been systematically addressed across the product lifecycle. Those foundations are essential. But they are not sufficient on their own.
Risk management is not static. It must adapt to the lifecycle phase of the product.
Two phases consistently concentrate on the highest level of uncertainty, learning, and exposure: initial product launch and product change or major modification. Treating these phases the same way we treat mature, stable manufacturing is one of the most common and most costly risk management mistakes in MedTech.
This article focuses on why these phases are different, what risk management activities must change, and how the automotive concept of Safe Launch could become a game changer if thoughtfully adapted to MedTech.
Why Launch and Change Are Fundamentally Different from Mature Manufacturing
Mature manufacturing operates in a data-rich environment. Processes are characterized, failure modes are well understood, suppliers are stable, and feedback loops are predictable. Risk management in this phase is largely about control and surveillance.
Initial launch and product change operate in the opposite conditions.
At launch, assumptions are still being tested against reality. At change, previously validated assumptions may no longer be true. In both cases, the organization is exposed to unknown unknowns, not just residual risks documented in a file.
Applying steady-state controls to these phases creates a false sense of security. The risk management file may look complete, but the operational reality has shifted.
Lifecycle Phase 1: Initial Product Launch
What Makes Launch High Risk
Initial product launch is the first time a design, a manufacturing process, and a supply chain interact at scale under real-world conditions. Even with robust design controls, this phase includes:
Limited production history
Early learning curves in manufacturing and inspection
Real-world use conditions that differ from validation scenarios
Immature feedback loops from complaints and service
From a risk perspective, launch is where latent risks surface.
Risk Management Activities That Must Be Intensified
During initial launch, risk management should shift from document completeness to behavioral vigilance. Key techniques typically include:
Reconfirmation of DFMEA and PFMEA assumptions under production conditions
Temporary tightening of process controls and inspection criteria
Increased sampling, monitoring, and trend review frequency
Rapid escalation paths for deviations and near-misses
Explicit linkage between early complaints, nonconformances, and risk evaluation
The goal is not to redesign the product at launch, but to detect weak signals before they become patient harm or regulatory findings.
Lifecycle Phase 2: Product Change or Major Modification
Why Product Changes Are Often Underestimated
Product changes often occur in mature systems, which creates dangerous bias. Because the product has a history, teams assume risk is inherently lower.
In reality, a product change can invalidate years of accumulated knowledge.
Changes to materials, suppliers, software, tolerances, processes, labeling, or sterilization can reintroduce hazards that were previously controlled or irrelevant.
Risk Management Activities for Product Changes
Effective risk management during product change focuses on delta thinking, not full repetition. Typical activities include:
Change-specific risk analysis focused on what is different.
Targeted DFMEA and PFMEA updates tied to the change scope.
Impact analysis across design, manufacturing, suppliers, and post-market activities
Verification that prior risk acceptability decisions still apply.
Heightened monitoring immediately after change implementation
The goal is to prevent risk from silently re-entering a system that appears stable on paper.
A Comparative View of Risk Profiles Across Lifecycle Phases
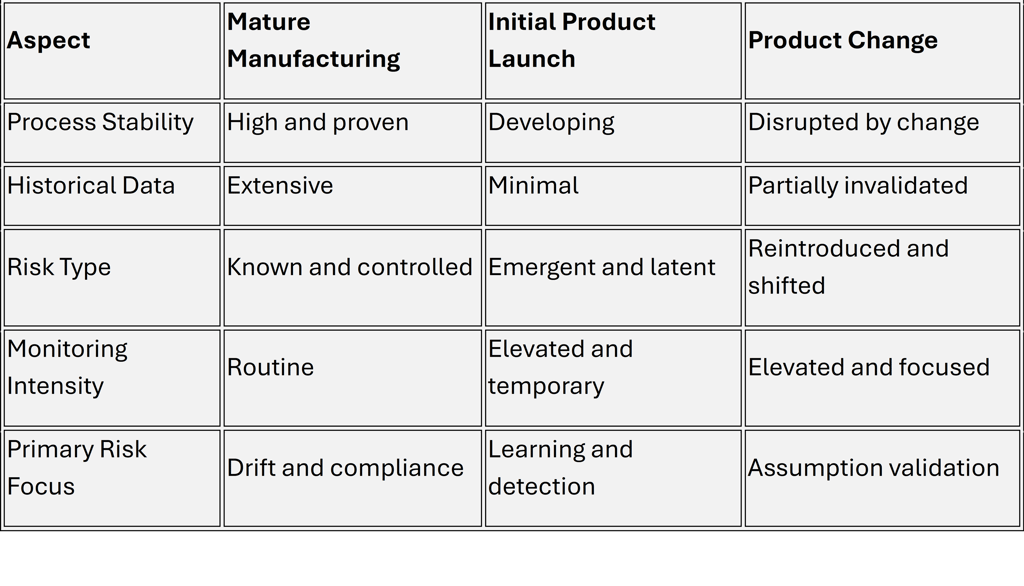
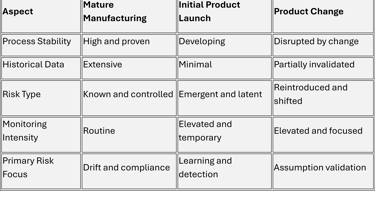
This comparison highlights why a single, uniform risk management approach across all phases is ineffective.
Introducing the Automotive Safe Launch Concept
In the automotive industry, Safe Launch is a structured, temporary operating mode used during product launch or major change. It acknowledges a simple truth: early production is not business as usual.
Safe Launch typically includes:
Temporarily heightened controls.
Increased inspection and verification
Explicit management oversight
Rapid feedback and escalation mechanisms
Clear criteria for exiting Safe Launch
It is not a tool. It is a risk posture.
Why Safe Launch Is a Game Changer for MedTech
MedTech already recognizes lifecycle risk in theory. ISO 14971 explicitly requires risk management throughout the product lifecycle. What is often missing is an operational mechanism that reflects changing risk profiles.
A MedTech-adapted Safe Launch approach bridges that gap.
It provides a structured way to align operational behavior with documented risk assumptions during periods of maximum uncertainty. It strengthens patient safety without relying on reactive CAPA. And it demonstrates to regulators that the organization understands not just risk documentation, but risk dynamics.
What a MedTech Safe Launch Could Look Like
A MedTech Safe Launch does not need new regulations or new standards. It requires intentional execution. A practical framework might include:
A defined Safe Launch period tied to production volume or time.
Temporarily elevated inspection, monitoring, and review cadence
Clear ownership across Quality, Operations, and Engineering
Explicit escalation rules tied to predefined risk signals.
Objective exit criteria that return the product to normal operations
Importantly, Safe Launch applies equally to new products and high-impact product changes.
Key Takeaways
Risk management must adapt to product lifecycle phases, not remain static.
Initial launch and product change represent peak uncertainty and exposure.
Mature manufacturing controls are insufficient during these phases.
The automotive Safe Launch concept offers a proven, adaptable model.
A MedTech Safe Launch bridges the gap between risk planning and real-world execution.
In modern, lifecycle-aware risk management, success is not defined by how complete the file looks, but by how effectively the organization behaves when uncertainty is highest.
Why NoioMed Is Uniquely Positioned to Enable Safe Launch in MedTech
Adapting a Safe Launch concept from automotive into MedTech requires more than copying tools or checklists. It demands deep understanding of both industries, their regulatory expectations, and how risk actually manifests on the manufacturing floor and in the field.
NoioMed was founded specifically to work at that intersection.
Under the leadership of its founder, Manfred Maiers, NoioMed brings decades of hands-on experience across regulated MedTech environments and high-risk automotive applications, including off-road and extreme-use platforms where Safe Launch principles are not optional but essential for survival.
This dual-industry background matters.
Safe Launch is not about adding more paperwork. It is about translating risk awareness into disciplined execution during periods of uncertainty. NoioMed understands how to:
Align Safe Launch behaviors with ISO 14971 and FDA QMSR expectations.
Integrate launch and change-phase risk controls into existing QMS structures.
Bridge Quality, Engineering, and Operations without creating parallel systems.
Design temporary, phase-specific controls that strengthen safety without slowing innovation.
Most importantly, NoioMed approaches Safe Launch as a lifecycle capability, not a one-time event. Whether supporting first product commercialization or managing high-impact product changes, the focus stays the same: protecting patients, preserving regulatory confidence, and enabling organizations to learn faster than their risks evolve.
In an industry where uncertainty is inevitable, Safe Launch becomes a discipline. With its cross-industry perspective and deep MedTech credibility, NoioMed is positioned to help make that discipline practical, scalable, and regulator-ready.
