Functional Safety in MedTech
When IEC 61508 Thinking Helps, When It Hurts, and Where Hybrid Models Win.
RISK MANAGEMENTTHE LEARNING LOOP
Manfred Maiers
3/5/20266 min read


Functional Safety in MedTech
When IEC 61508 Thinking Helps, When It Hurts, and Where Hybrid Models Win.
Medical devices are no longer “just” devices. They are increasingly systems: software-controlled, sensor-rich, network-connected, and often operating in tight feedback loops with the patient.
That evolution is forcing a new question that many MedTech teams are starting to ask quietly, and a few are starting to ask out loud:
If industries like aerospace, automotive, rail, and industrial automation rely on functional safety frameworks like IEC 61508 to engineer safety into complex systems, should medical devices do the same?
And just as important:
Does this thinking apply only to complex electromechanical systems like infusion pumps and surgical robots, or does it belong anywhere near disposables like needles, tubing, and infusion sets?
This article takes a practical look at what functional safety really is, where IEC 61508 style methods bring real value in MedTech, where they are unnecessary overhead, and how hybrid models can give you the best of both worlds.
What functional safety actually means
Functional safety is not “more risk management.” It is a specific engineering discipline for preventing harm caused by a system not performing its safety-related functions correctly.
At its core, functional safety asks:
What safety function must the system perform to prevent unacceptable harm?
What failure modes could prevent that function?
How likely are those failures, including random hardware failures and systematic failures (design, software, process).
What architecture, diagnostics, redundancy, and verification are required so that safety function performance is achieved with a defined confidence.
IEC 61508 is the foundational cross-industry standard that formalizes this approach. It introduces concepts that are less emphasized in typical MedTech implementations of ISO 14971, such as:
Safety Integrity Levels (SIL) to express required risk reduction and confidence targets.
Quantitative reliability modeling for random hardware failures.
Systematic capability expectations around lifecycle rigor, tools, and verification.
Architectural constraints such as redundancy and fault tolerance.
Diagnostic coverage as a first-class design property, not an afterthought.
You do not need to “be IEC 61508 compliant” to benefit from these ideas, but you do need to understand what they are designed to solve.
How medical device risk management works today
Most MedTech companies manage risk primarily under ISO 14971. Done well, this is a solid framework. It provides a structured way to:
Identify hazards and hazardous situations.
Estimate and evaluate risks.
Implement risk controls.
Verify risk controls and assess residual risk.
Maintain risk acceptability across the lifecycle.
In practice, teams typically use tools like:
DFMEA and PFMEA.
Hazard analysis and use-related risk analysis.
Fault tree analysis for select hazards.
Verification and validation evidence tied back to controls.
Where ISO 14971 is sometimes weaker in day-to-day execution is not the standard itself, but how organizations apply it to highly complex, software-driven, dynamic systems. Many teams remain largely qualitative, and many hazard controls are treated as requirements rather than engineered safety functions with measurable integrity.
That is precisely the gap functional safety was built to address.
Where functional safety thinking adds value in MedTech
Functional safety shines where harm prevention depends on correct system behavior, especially when:
Software logic is safety-relevant.
Sensors, actuators, and feedback loops can fail silently.
A single failure can produce immediate harm.
Device behavior changes dynamically during operation.
Architecture decisions like redundancy and diagnostics meaningfully reduce risk.
This maps well to devices such as:
Infusion pumps and automated drug delivery systems.
Surgical robotics.
Radiation therapy delivery systems.
Implantable active devices with embedded firmware.
Closed-loop therapy systems.
Devices with patient-connected automation and remote updates.
Functional safety methods help answer questions ISO 14971 teams often struggle to quantify:
What is the probability of a dangerous failure per hour?
What diagnostic coverage is actually achieved?
What happens when a sensor drifts but stays “in range.”
What independent monitoring is needed to detect systematic failures?
How much safety margin is created by architecture, not just requirements.
Where functional safety is usually overkill
There are many medical devices where IEC 61508 style rigor provides little incremental safety benefit, because risk is dominated by:
Material properties and geometry.
Manufacturing variation and process capability.
Sterility and packaging integrity.
Simple mechanical misuse scenarios.
Human factors and labeling.
This includes many disposables and passive devices:
Needles.
Infusion sets and tubing sets.
Catheters.
Syringes.
Simple connectors and valves.
Wound dressings and basic mechanical disposables.
The risks are real, but the failure physics are typically not “safety function integrity” problems. They are design robustness, process control, usability, and biocompatibility problems.
Imposing IEC 61508 lifecycle structure here can create heavy documentation and verification overhead without improving patient safety proportionally.
Traditional ISO 14971 vs functional safety frameworks
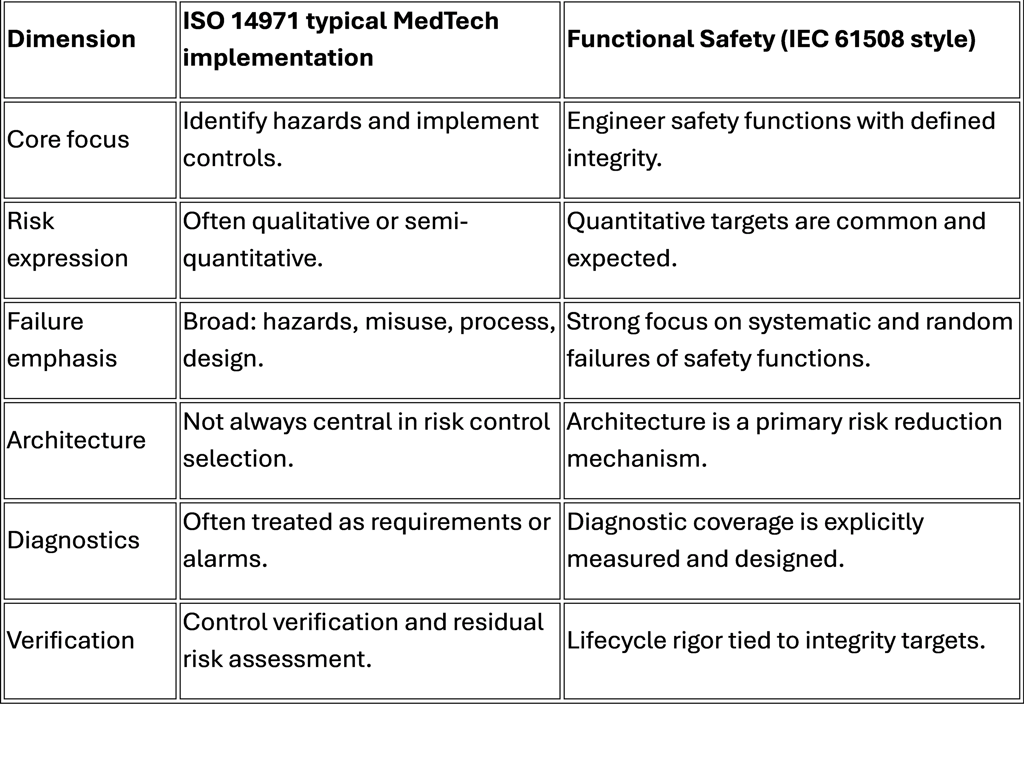
Cost vs value
The cost of adopting functional safety frameworks is not just training or templates. It changes how engineering is performed:
Deeper system decomposition into safety functions.
Formal safety requirements and allocation.
Reliability models and failure rate assumptions.
Expanded verification, including fault injection strategies.
Tool qualification and lifecycle process controls in some cases.
This is absolutely justified in some product classes. It is wasteful in others.
The right question is not “Should we implement IEC 61508.”
The right question is:
Which functional safety elements reduce our top patient harm risks more effectively than our current ISO 14971 execution, and at what cost?
Hybrid model opportunities
Most MedTech organizations do not need to fully adopt IEC 61508 to benefit from it.
Hybrid approaches are often the sweet spot. Examples of functional safety elements that map well into MedTech without forcing a full framework migration:
Safety function definition and allocation for high-severity hazards.
Architecture-level fault detection and safe-state behavior.
Diagnostic coverage thinking for sensors and actuators.
Quantitative reliability modeling for selected hazard chains.
Strong separation between control logic and independent monitoring.
Formal verification for safety-critical software paths.
Hybrid models also reduce risk of over-processing. You apply functional safety rigor only where it meaningfully improves the integrity of harm-preventing behavior.
Small case example
Infusion pump vs disposable infusion set
This comparison is intentionally simple, but it illustrates why “functional safety everywhere” is not a good strategy.
Case A: Infusion pump (complex electromechanical system)
Context
An infusion pump is a software-controlled electromechanical system delivering medication over time. Harm can occur quickly if delivery rate is wrong, if occlusion is missed, or if software or sensor faults create undetected deviations.
Representative high-severity hazardous situation
Over-infusion leading to overdose.
What ISO 14971 teams often do well
Identify overdose hazard and define controls such as flow monitoring, alarms, user confirmations, and verification testing.
Create DFMEA items for pump mechanism, sensors, software, and user interaction.
Add labeling, training, and use instructions.
Where functional safety thinking can meaningfully improve outcomes
Define the safety function explicitly: “Prevent delivery outside specified bounds or detect and stop within X seconds.”
Allocate that function across architecture: primary control loop plus independent monitoring channel.
Establish integrity expectations: probability of dangerous failure per hour and diagnostic coverage requirements.
Analyze systematic failures: software logic errors, sensor plausibility errors, update or configuration hazards.
Validate safe-state behavior: pump stop, alarm escalation, lockout strategies.
Use fault injection and negative testing to verify detection capability, not just normal operation.
Net result
Functional safety elements can convert a loosely defined “alarm requirement” into a designed safety mechanism with measurable integrity, especially when software and sensors dominate risk.
Case B: Disposable infusion set (simple device)
Context
A disposable infusion set is largely passive. Its safety risks typically arise from mechanical integrity, flow restriction, leakage, misconnections, sterility, and use error. There is no software control loop, and architecture-level redundancy is usually not meaningful.
Representative high-severity hazardous situation
Leakage or occlusion leading to under-delivery or infiltration.
What ISO 14971 teams often do well
Identify leakage, occlusion, disconnection, and misconnections.
Apply controls such as material choice, dimensional tolerancing, connector standards, packaging integrity, usability labeling, and manufacturing process controls.
Verify via burst pressure tests, leak tests, flow tests, tensile strength, and aging studies.
Tie risks strongly to process capability, incoming inspection, and supplier controls.
Why functional safety provides little incremental value
There is no safety-related control logic to engineer for integrity.
“Diagnostic coverage” does not apply in the same way, because the device is not monitoring itself.
Random hardware failure modeling is less relevant than process variation and material defects.
The most powerful controls are design robustness and manufacturing control, not redundant architecture or safe-state logic.
Net result
IEC 61508 style processes would add lifecycle overhead without improving the primary risk drivers. A stronger PFMEA, supplier controls, and process validation strategy often deliver more safety per dollar.
Decision guide
When functional safety thinking adds value
Functional safety concepts are generally worth considering when:
A hazard is driven by software, sensors, or control algorithms.
A failure can be silent and rapidly harmful.
You need engineered detection and safe-state response.
Architecture choices can materially reduce risk.
The device operates continuously over time with dynamic control
Functional safety concepts are generally not worth the overhead when:
The device is passive and failures are visible or purely mechanical.
Risk is dominated by manufacturing variation, materials, sterility, and packaging.
Controls are primarily process and inspection driven rather than system behavior driven.
Closing insight
MedTech does not need to abandon ISO 14971. It is the right backbone for medical device risk management.
But for modern devices that behave like safety-critical systems, ISO 14971 alone can become too qualitative unless it is executed with stronger engineering discipline.
The most practical path for many companies is a hybrid approach: keep ISO 14971 as the governing framework and selectively import functional safety concepts where integrity of system behavior is the real risk driver.
That is where the biggest opportunity sits: not in adopting IEC 61508 as a checkbox, but in borrowing its best engineering mechanisms to make risk controls more robust, measurable, and resilient.
