Enhancing PFMEA with Operational Excellence Attributes
PFMEA beyond Risk Management in MedTech Manufacturing
AI-ENHANCED OPERATIONAL EXCELLENCETHE LEARNING LOOP
Manfred Maiers
9/12/20254 min read


Enhancing PFMEA with Operational Excellence Attributes: Beyond Risk Management in MedTech Manufacturing
Introduction and Article Overview
Failure Mode and Effects Analysis (FMEA) is a cornerstone in MedTech risk management, traditionally applied through Design FMEA (DFMEA) and Process FMEA (PFMEA). While DFMEA ensures safe and robust product designs, PFMEA focuses on the manufacturing and assembly processes that bring those designs to life.
Historically, PFMEA has been used primarily as a compliance and risk management tool, centered on severity, occurrence, and detection ratings. While this framework protects patients and users, it often overlooks the operational and financial dimensions of process failures. In an era of rising cost pressures, supply chain volatility, and increasing regulatory scrutiny, this narrow use of PFMEA represents a missed opportunity.
This article explores how PFMEA can be enhanced with new attributes that extend its value beyond risk management to support operational excellence. By integrating cost-of-quality and process impact considerations into PFMEA, organizations can achieve a balanced view that protects patients, satisfies regulators, and strengthens business performance.
The Basics of FMEAs: DFMEA vs. PFMEA
Failure Mode and Effects Analysis (FMEA) is a structured approach to identifying, evaluating, and mitigating risks that could result in product or process failures.
- Design FMEA (DFMEA): Focuses on potential failure modes in the design phase of a medical device. Its primary goal is to prevent design weaknesses that could impact patient safety, regulatory compliance, or long-term product reliability.
- Process FMEA (PFMEA): Focuses on potential failures that could occur during manufacturing and assembly processes. PFMEA evaluates how process variations, operator errors, or equipment malfunctions could create nonconformities or risks to the final device.
Together, DFMEA and PFMEA form a continuum of risk management: DFMEA ensures robust product design, while PFMEA ensures robust and repeatable manufacturing processes.
PFMEA in Manufacturing and the Role of RPN
In production, the PFMEA plays a critical role in ensuring both product safety and regulatory compliance. The Risk Priority Number (RPN) remains the traditional metric used to evaluate risk:
- Severity (S): How serious the consequence of the failure would be for the patient, user, or system.
- Occurrence (O): The probability that the failure will happen.
- Detection (D): The likelihood that the failure will be detected before reaching the patient or user.
RPN is calculated as: RPN = S × O × D
The PFMEA’s ultimate purpose in MedTech is patient and user safety. It ensures that risks are systematically reduced to an acceptable level, meeting both FDA and ISO 13485 requirements. However, while safety is paramount, this framework often overlooks another critical element: operational efficiency and the cost of quality.
The Problem with Detection in FMEAs
A key issue in traditional FMEAs is how “detection” is treated. In many cases, if a failure mode has a high detection rating (e.g., reliable inspection or testing catches the issue), the residual risk is considered acceptable and no further mitigation is required.
From a regulatory standpoint, this makes sense: the patient or user is protected. However, from an operations and quality standpoint, this approach hides significant inefficiencies and costs:
- Rework: If an error is caught mid-process, rework may be required, consuming time and labor.
- Scrap: If the error is caught late—such as during final inspection—the entire device may need to be scrapped, resulting in material and revenue loss.
- Supplier Corrective Action Requests (SCARs): Errors traced back to suppliers may trigger formal corrective actions, adding administrative burden and potential supply chain risk.
Example: Wrong Material in Manufacturing
- Detected at Incoming Inspection: Nonconforming material is quarantined, SCAR is issued, production continues with minimal disruption. Cost impact: moderate.
- Detected at Final Inspection: Device cannot be reworked, must be scrapped. Cost impact: severe.
Thus, while detection reduces patient risk, the operational and financial impact varies dramatically depending on when the failure mode is detected.
Extending PFMEA with Cost and Operational Attributes
To align safety-driven risk management with operational excellence, PFMEAs can be enhanced by adding cost and process impact attributes alongside the traditional S–O–D ratings.
Proposed New Attributes:
1. Impact Type: Categorize the operational consequence (Rework, Scrap, SCAR, Line Stop).
2. Cost of Failure (Before Mitigation): Estimated direct and indirect costs if the failure occurs and is detected late.
3. Cost of Failure (After Mitigation): Residual cost when controls are applied.
4. Cost Avoidance Potential: The difference between before- and after-mitigation costs, enabling prioritization by both safety risk and financial impact.
Application to Risk Graphs in PFMEA
Some PFMEAs use risk graphs instead of RPN to assess process-related risk. These graphs typically evaluate severity, frequency, and probability of avoidance. While effective in safety-critical contexts, they also lack visibility into operational cost impact.
Just as RPN can be extended with cost-of-quality attributes, risk graphs in PFMEAs can be augmented with:
- Impact Type (Rework, Scrap, SCAR, Line Stop)
- Cost-of-Quality Metrics (before and after mitigation)
This provides a dual lens: one for safety compliance and one for operational excellence.
Conclusion and Benefits Analysis
By extending PFMEA with cost and operational attributes, organizations can transform a compliance-driven risk management tool into a strategic enabler of operational excellence.
Key Benefits:
- Improved Decision-Making: Prioritize mitigations not only by safety risk, but also by cost and revenue impact.
- Cost of Quality Reduction: Identify opportunities to prevent rework, scrap, and supplier issues before they occur.
- Regulatory Alignment: Demonstrate proactive integration of quality, regulatory, and operational perspectives.
- Operational Excellence: Bridge the gap between process risk management and continuous improvement.
The enhanced PFMEA is not just a defensive tool for compliance—it becomes an offensive tool for operational excellence, aligning MedTech organizations with both regulatory expectations and business success.
Visual Frameworks
The following visuals illustrate the evolution from traditional PFMEA to enhanced PFMEA and highlight the added attributes:
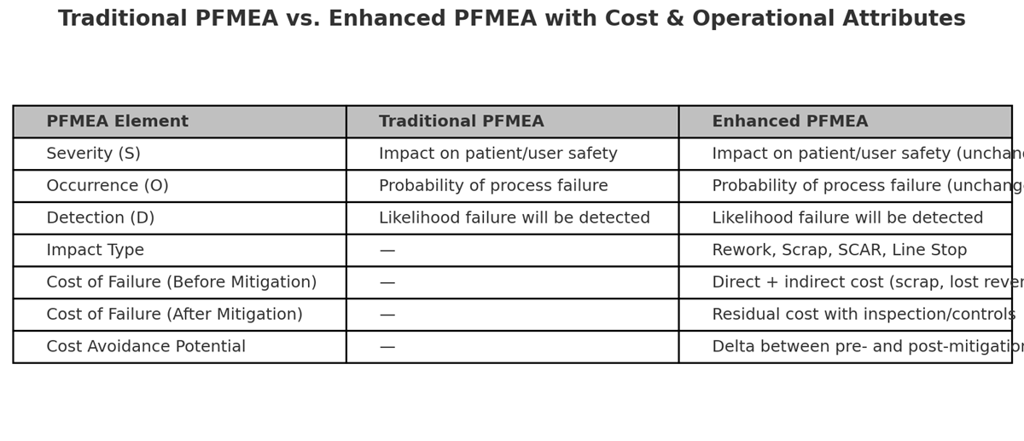
Figure 1: Traditional PFMEA vs. Enhanced PFMEA Comparison Table
